 If you say a mole of donuts, that means you have 6.022 x 10 23 donuts and a very bad stomach ache. So much in the same way you would say a dozen donuts and know that meant 12 donuts. While I know that does not make much sense right now, the important thing to know is that the value of the mole is 6.022 x 10 23entities. The official definition of the mole is that it is an amount of entities equal to the number of atoms in 0.012 grams of Carbon 12. The mole is a very important unit in chemistry and one we will be using in the near future A LOT. The last two SI base units we will discuss are those for the amount of a substance, the mole and the unit for luminous intensity ( a fancy way of saying how bright a light might be) called the Candela (I always think of candle light to help me remember this one). 
The kelvin, unit of thermodynamic temperature, is the fraction 1/273.16 of the thermodynamic temperature of the triple point of water. The ampere is that constant current which, if maintained in two straight parallel conductors of infinite length, of negligible circular cross-section, and placed 1 meter apart in vacuum, would produce between these conductors a force equal to 2 x 10 -7 newton per meter of length. The Fahrenheit degree scale is not equivalent to the kelvin scale so further calculations must be completed to convert between those two temperature scales. Therefore to convert from degrees Celsius to kelvin, you simply add 273.15 to the Celsius temperature. On the kelvin scale, absolute zero is 0 K and the triple point of water has the value of approximately -273.15 degrees Celsius. The kelvin measurement intervals are equal to those of the Celsius degree scale. Kelvin is called the absolute temperature scale because it does not use degrees. 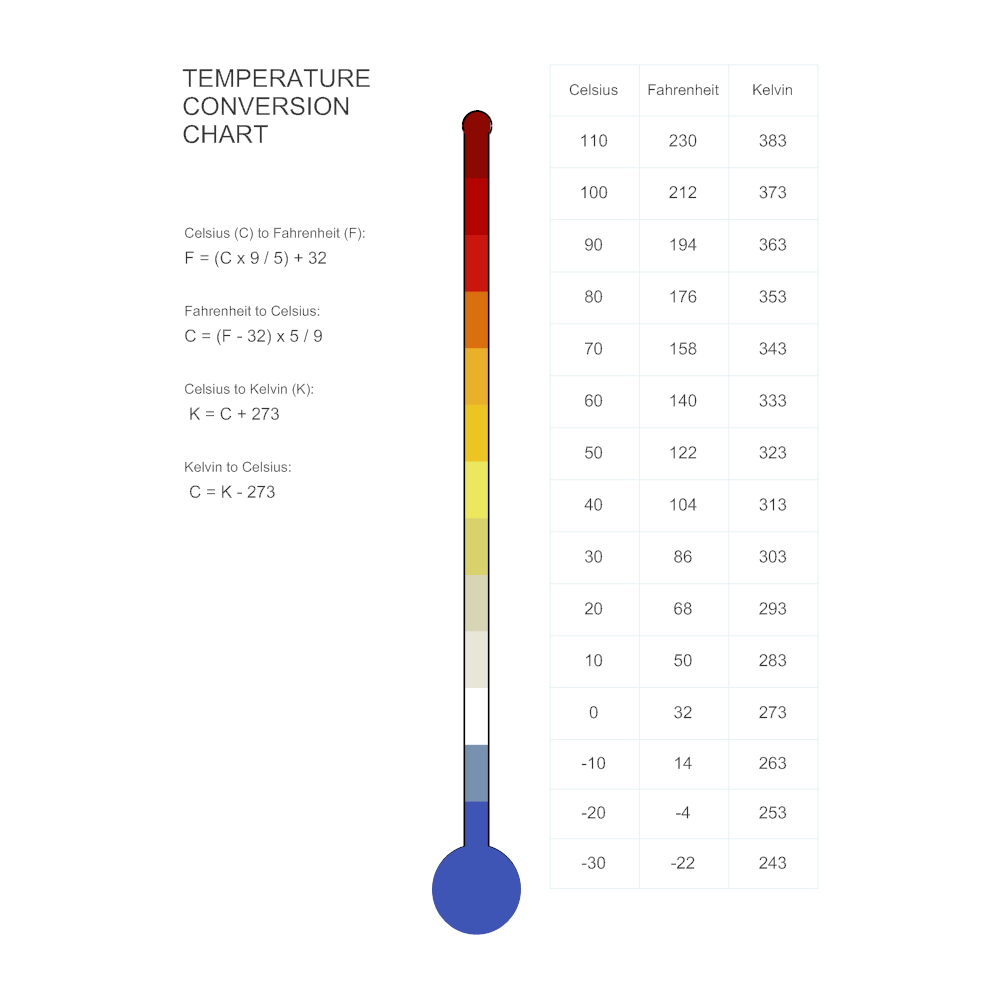
The kelvin (K) is the base unit for temperature. Generally speaking both units stand for a package of electrical current that has a force equal to that of 2 x 10 7 newtons per meter of length. A Coulomb is in turn defined as 1 Ampere per second. The Ampere is that constant current which travels at a rate of 1 coulomb per second. While we won’t use the unit for electric current often, it is good to know the basis for the unit. The base units for Electric Current and temperature are the Ampere and the Kelvin, respectively. The second is the duration of 9,192, 631, 770 periods of the radiation corresponding to the transition between the two hyperfine levels of the ground state of the cesium 133 atom. The kilogram is the unit of mass it is equal to the mass of the international prototype of the kilogram. The meter is the length of the path travelled by light in vacuum during a time interval of 1/299,792,458 of a second. As for time, although you might think the minute a better unit, the reality is that most chemical reactions happen very quickly and a minute is too large a unit to be very handy for expressing the time scale of these reactions. I would much rather weigh 59 kg than 59,000g. The reason the kilogram is used for the unit of mass is because the gram is such a small amount. There are in fact 2.2 lbs in a kilogram if that will help you picture it better. While the meter should be quite familiar to you, as meter sticks are slowly becoming as common as yard sticks in this country, the kilogram is probably not an amount or mass that you can picture readily in your head like you could a pound. Three of the most commonly used units are those for length, mass and time. You should definitely familiarize yourself with the units, their symbols and what measurement type they represent. The table presented here lists all of the common SI base units. 
In chemistry the scientists have come to agree upon a set of common units called the System de International (SI) base units that we all agreed to use when communicating our research findings. This ability however is highly dependent on the units in which the data is expressed. As was mentioned previously, one of the more important aspects of quantitative data is the ability to use it for comparison. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
